
This celestial scene represents the work by Savani et al. to track the constellation of metabolites that contribute to cellular nitrogen metabolism. Artwork by Melissa Logies.
McBrayer Lab uses their new nitrogen metabolism profiling platform, organoid model to discover modification of enzyme CAD activates de novo pathway
A cell’s development stage intrinsically determines its use of de novo and salvage pathways to synthesize pyrimidine nucleotides – building blocks of DNA required for cellular growth and function – a discovery made using a newly-developed nitrogen metabolism mapping system built by Children’s Medical Center Research Institutive at UT Southwestern (CRI) researchers, per a study published today in Nature Metabolism.
How cells use nutrients containing nitrogen is less studied than those with carbon because of the many different sources of nitrogen available. To address this challenge, CRI scientists created a library of dozens of human plasma-like medium (HPLM) stocks, each with a distinct metabolite to track nitrogen. They discovered immature, primitive cells use both pathways but primarily the de novo, from scratch, pathway, while mature, differentiated cells relied on uridine pools, which are partially-formed building blocks recycled in the salvage pathway.

Sam McBrayer, Ph.D., (left), Assistant Professor in CRI and of Pediatrics, watches as Milan Savani, Ph.D., loads samples at CRI’s Metabolomics Shared Facility.
The study was led by Sam McBrayer, Ph.D., Assistant Professor in CRI and of Pediatrics, and co-senior author Kalil Abdullah, M.D., a neurosurgeon and Associate Professor at University of Pittsburgh, in addition to researchers from both labs.
In the brain, neural stem cells are primitive, or immature, before they age and differentiate into neurons or glial cells. Glia are often called “the glue” in brain anatomy because they physically surround and support neurons. One type of glial cell is an astrocyte, the star-like cells that directly surround a neuron. When glial cells become dysregulated, they can become tumors, called gliomas.
In this study, first author Milan Savani, Ph.D., a student in UT Southwestern’s Perot Family Scholars Medical Scientist Training Program and McBrayer Lab researcher, compared mature, non‑malignant astrocytes with primitive, malignant glioma cells. His research showed glioma cells favored de novo pyrimidine synthesis, while astrocytes relied on uridine salvage.
Dr. Savani and researchers then tracked normal brain cells and cells from multiple human tumors during uridine deprivation experiments, cutting the nutrient supply to the salvage pathway.
Scientists found that modification of a region called S1900 on the enzyme CAD operates as a “switch” to turn on the de novo pathway in mature cells when nutrients for the salvage pathway are limited. In addition, the CAD S1900 switch could be triggered in mature cells, even when uridine was abundant, to reproduce this metabolic feature of primitive cells.
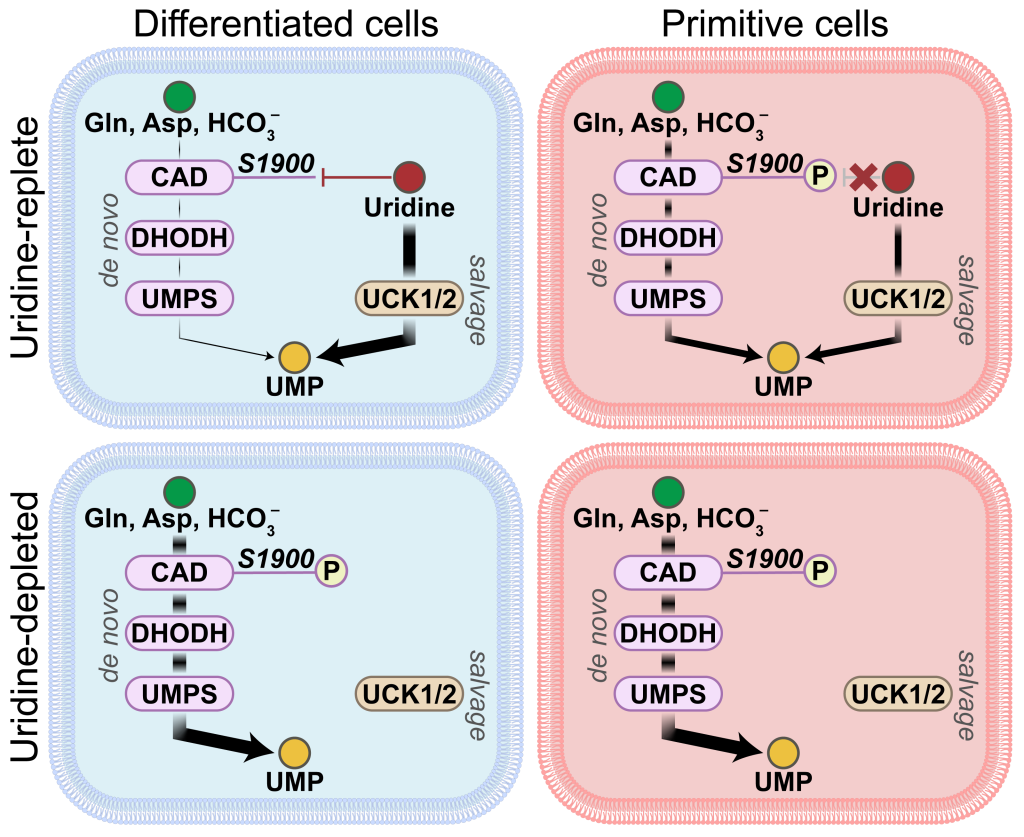 “Independent of nutrient availability, this difference is intrinsic to cell identity and linked to cell differentiation state rather than enzyme expression level,” Dr. McBrayer said.
“Independent of nutrient availability, this difference is intrinsic to cell identity and linked to cell differentiation state rather than enzyme expression level,” Dr. McBrayer said.
This latest research builds on an organoid model the McBrayer Lab created, which they adapted last year using HPLM to make amenable to metabolic mapping studies while preserving the viability and key biological features of the surrounding microenvironment cells after brain tumors are resected.
“We know there’s crosstalk between tumor cells and the neighboring cells in cancers. And capturing those interactions is very important,” Dr. McBrayer said. “This study shows how organoid models provide a more physiologically relevant context to study metabolic interactions between cells in brain tumors. Prior to this work, it has been difficult to study these relationships using human tumors that were just snap-frozen.”
This recent discovery continues to refine why some gliomas are hyperdependent on de novo pyrimidine nucleotide synthesis, a 2022 McBrayer Lab discovery published in Cancer Cell.
“The molecular switch seems to be stuck in ‘build’ mode in some cancers and may represent a therapeutic target in certain brain tumors,” Dr. Savani said. “Since healthy, mature cells prefer to recycle, they may be less affected by these treatments. This enzyme modification could also serve as a biomarker that allows us to predict which tumors will respond to these therapies.”
Dr. McBrayer is a Cancer Prevention and Research Institute of Texas (CPRIT) Scholar and winner a Distinguished Scientist Award in 2021 from the Sontag Foundation. He is also a member of the Cellular Networks in Program of the Harold C. Simmons Comprehensive Cancer Center at UT Southwestern.
Dr. Savani is currently finishing his M.D. at UT Southwestern before moving on to a residency program next year. His work was supported by a Ruth L. Kirschstein National Research Service Award from the National Cancer Institute, and he was recently awarded the Leita Marsh Award, sponsored by Altrusa International, Inc., for his work on these discoveries.
This research was funded by the National Institutes of Health, Oligo Nation, CPRIT, American Heart Association, and the Human Frontier Science Program.
###
About CRI
Children’s Medical Center Research Institute at UT Southwestern (CRI) is a joint venture of UT Southwestern Medical Center and Children’s Medical Center Dallas. CRI’s mission is to perform transformative biomedical research to better understand the biological basis of disease. Located in Dallas, Texas, CRI is home to interdisciplinary groups of scientists and physicians pursuing research at the interface of regenerative medicine, cancer biology, and metabolism — relentless discovery toward the treatments of tomorrow.
X/Twitter | Blue Sky | LinkedIn | Instagram | YouTube | Facebook



