February 25, 2020
Brandon Faubert, Ph.D., is a Canadian Institutes of Health Research Fellow in Children’s Medical Center Research Institute (CRI) at UT Southwestern. He works in the DeBerardinis lab studying tumor metabolism. His work has the potential to uncover new therapies and imaging techniques for patients suffering from lung cancer – the leading cause of cancer deaths worldwide.
What are you researching?
Metabolism is important for the survival of all living things, including tumors. Our group is interested in how and why tumors use certain nutrients to survive and grow. There have been numerous discoveries involving the altered metabolism of cancer cells grown in a dish but that is only part of the picture. Until recently, we’ve lacked an understanding about the metabolism of tumors in patients. I’ve been fortunate enough to be involved in a clinical study that examines tumor metabolism directly in patients with non-small cell lung cancer. This project is part of a large, multidisciplined collaboration between the clinic and bench led by Dr. Kemp Kernstine, Dr. Robert Lenkinski, and my mentor, Dr. Ralph DeBerardinis.
In the study, which is still ongoing, we combined magnetic resonance imaging and intraoperative 13C-nutrient tracing (a safe, non-radioactive isotope) to study how tumors use different metabolites and what pathways they fuel. We were also interested in insights we could gain from combining imaging techniques and metabolic analysis, which turned out to be a lot.

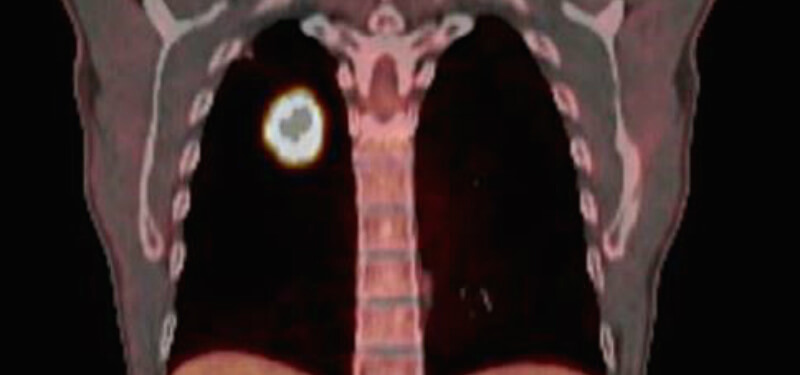
What have you discovered so far?
Our first discovery was how diverse tumor metabolism could be. Rather than a uniform mass of cells, tumors are remarkably complex. In a single tumor, regions that are millimeters apart can have a markedly different imaging and metabolic profile. Later, we discovered that lactate (traditionally thought of as a ‘waste product’ of cancer cells) functions as an important fuel source for some tumors. The study was published in Cell. Overall, this study has sparked new investigations into some exciting areas of cancer biology and collaborative projects across CRI.
How do you think your work will help patients?
Shortly after we published our lactate story, a postdoc in Sean Morrison’s laboratory, Aslan Tasdogan, approached us with the idea that tumor metabolism could be related to tumor metastasis. He found that their mouse models of melanoma had similar metabolism as our lung cancer patients, meaning that some of the tumors used lactate as a fuel. Specifically, he found that aggressive, highly metastatic melanomas used lactate as a fuel to help cells survive in the bloodstream. Importantly, he showed that if you use a drug to prevent lactate use, you stop the cancer cells from spreading. The paper was published in Nature last year.
Although this research was in mice, the drugs he tested are currently being used in cancer clinical trials. I am curious to see what these trials find, as our research suggests they may be useful for certain patients.


How did you end up in CRI?
I blame it all on Ralph. I collaborated with his laboratory several times during my Ph.D. training, and I knew that I wanted to pursue my postdoc fellowship with him. After joining his lab, the CRI became a serendipitous discovery. The people, science, environment, and collaboration here continuously impress me. I appreciate being able to work with people from a variety of disciplines. It’s easy to get tunnel vision during the daily grind of your own research project. Getting exposure to different ideas, techniques, or interpretations helped motivate my own work and led to some exciting new discoveries.
What do you like to do when you’re not in the lab?
I know this is going to perpetuate a Canadian stereotype, but I do enjoy playing hockey. There’s a large recreational hockey community in Dallas, and playing once or twice a week helps me stay sane. Otherwise, my girlfriend and I like to unplug from the city and go on camping and hiking trips throughout the state and country.
