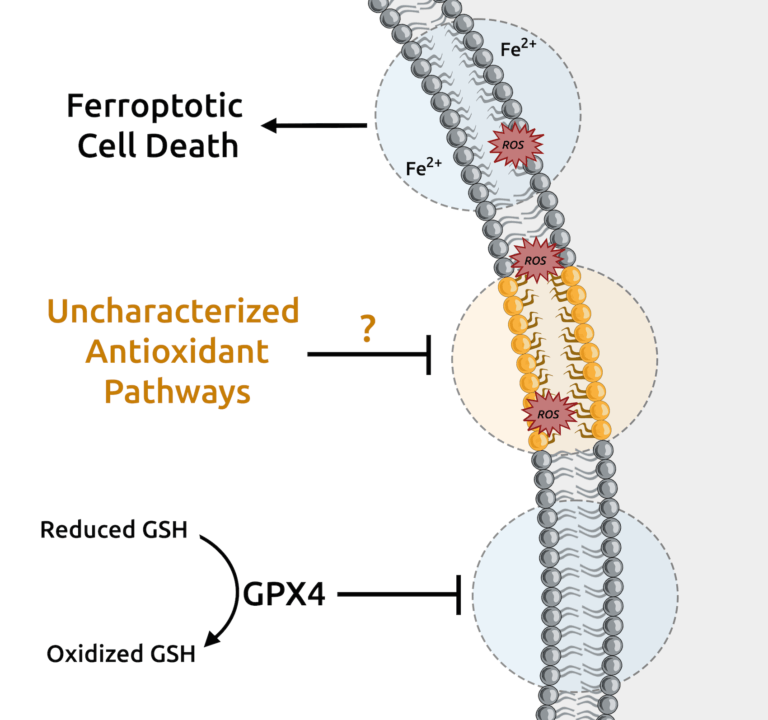
Calhoon, D.,# Sang, L.,# Ji, F., Bezwada, D., Hsu, S.C., Cai, F., Kim, N., Basu, A., Wu, R., Pimentel, A., Brooks, B., La, K., Paulina Serrano, A., Cassidy, D.L., Cai, L., Toffessi-Tcheuyap, V., Moussa, M.E., Uritboonthai, W., Hoang, L.T., Kolli, M., Jackson, B., Margulis, V., Siuzdak, G., Brugarolas, J., Corbin, I., Pratt, D.A., Weiss, R.J., DeBerardinis, R.J., Birsoy, K., and J. Garcia-Bermudez. (2025). Glycosaminoglycan-driven lipoprotein uptake protects tumours from ferroptosis. Nature 655, 799-808. (PubMed)
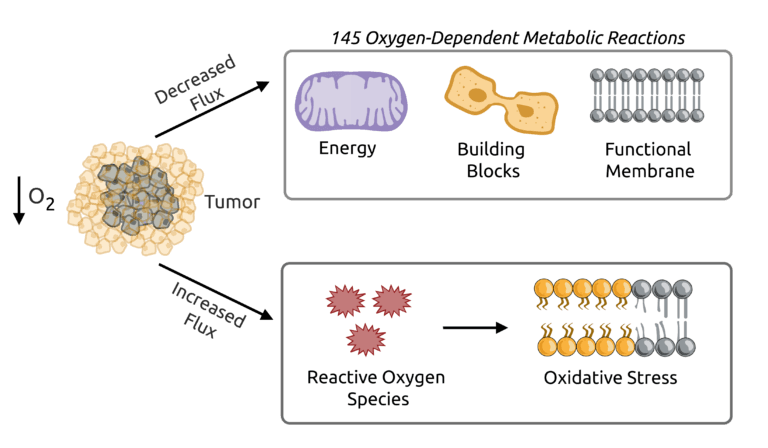
Garcia-Bermudez, J.,# Prasad, S.,# Baudrier, L., Badgley, M.A., Liu, Y., La, K., Soula, M., Williams, R.T., Yamaguchi, N., Hwang, R.F., Taylor, L.J., De Stanchina, E., Rostandy, B., Alwaseem, H., Molina, H., Bar-Sagi, D., and K. Birsoy. (2022). Adaptive stimulation of macropinocytosis overcomes aspartate limitation in cancer cells under hypoxia. Nature Metabolism 4, 724-738. (PubMed)
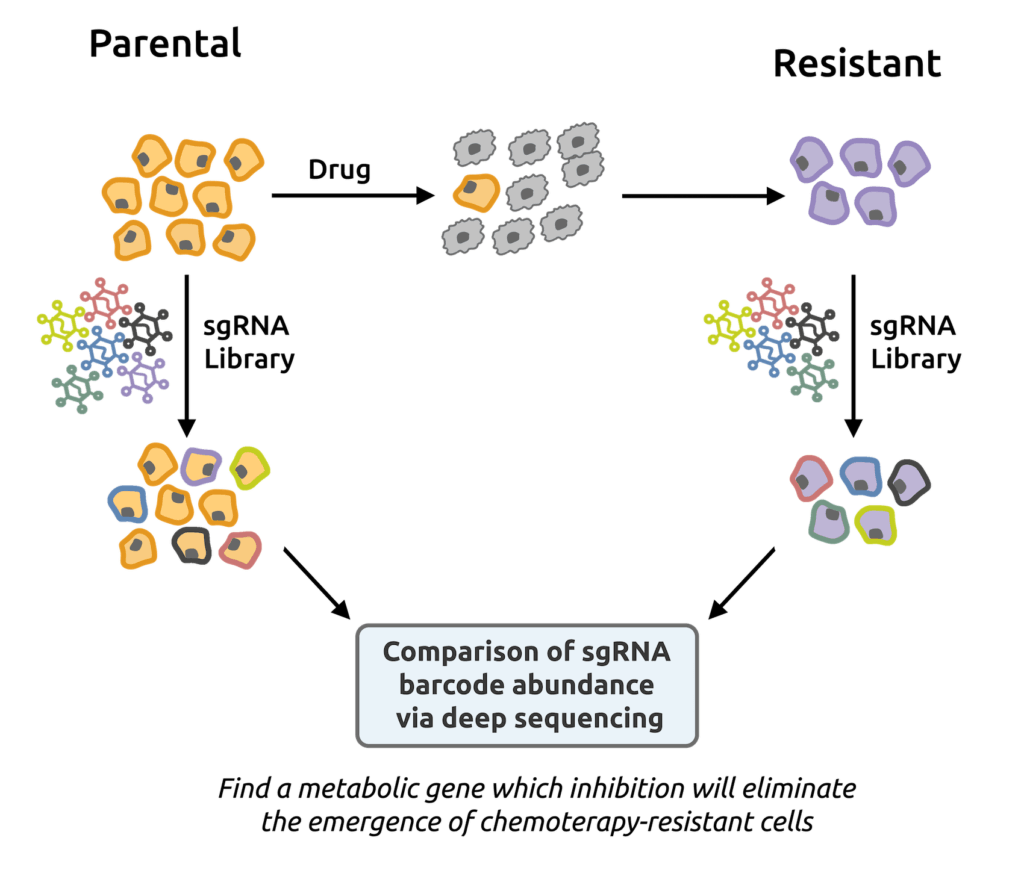
Soula, M., Weber, R., Zilka, O., Alwaseem, H., La, K., Yen, F., Molina, H., Pratt, D.*, Garcia-Bermudez, J.# and K. Birsoy.# (2020). Metabolic determinants of cancer cell sensitivity to canonical ferroptosis inducers. Nature Chemical Biology 16, 1351-1360. (PubMed)
Garcia-Bermudez, J., Baudrier, L., Bayraktar, E., Shen, Y., La, K., Guarecuco, R., Yucel, B., Fiore, S., Tavora, B., Freikman, E., Lewis, C., Min, W., Inghirami, G., La, K., Sabatini, D.M., and K. Birsoy. (2019). Squalene accumulation in cholesterol auxotrophic lymphomas prevents oxidative cell death. Nature 567, 118-122. (PubMed)
Garcia-Bermudez, J., La, K., Baudrier, L., Zhu, X.G., Sviderskiy, V.O., Papagiannakopoulos, T., Snuderl, M., Lewis, C., Possemato, R. and K. Birsoy. (2018). Aspartate is a limiting metabolite for cancer cell proliferation under hypoxia and in tumors. Nature Chemical Biology 20, 775-781. (PubMed)
García-Bermúdez, J., Sánchez-Aragó, M., Soldevilla, B., Del Arco, A., Nuevo-Tapioles, C., and J.M. Cuezva. (2015). PKA phosphorylates the ATPase Inhibitory Factor 1 and inactivates its capacity to bind and inhibit the mitochondrial H+-ATP synthase. Cell Reports 12, 2143-55. (PubMed)
#Co-corresponding author