

Shang Ma, Ph.D.
Shang Ma, Ph.D., received his bachelor’s degree from the University of British Columbia in Vancouver, Canada, with a major in genetics. He earned his Ph.D. in cellular and molecular biology from the University of Wisconsin in Madison, where he focused on chemical signaling pathways that mediate mammalian brain development. In 2015, Dr. Ma joined Dr. Ardem Patapoutian’s group at Scripps Research to study how cells sense mechanical forces during disease pathogenesis. During this postdoctoral training, he discovered that overactive PIEZO mechanosensitive ion channels play important roles in various human disorders.
In 2022, Dr. Ma joined the faculty of Children’s Medical Center Research Institute at UT Southwestern as an Assistant Professor. He holds a secondary appointment in the Department of Pediatrics.

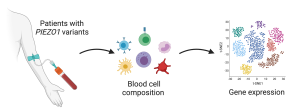
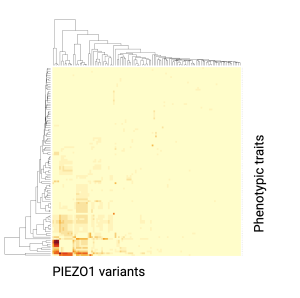 Rapid developments in precision medicine and computational science have provided us with a growing number of genome and metabolome databases to explore. Our lab is using these data to explore uncharted territories of human biology by creating exciting hypotheses to test. We have already found new mutations in genes encoding mechanosensitive proteins associated with surprising phenotypic traits from half a million individuals. We will investigate more novel areas of human biology where mechanotransduction plays unrecognized roles.
Rapid developments in precision medicine and computational science have provided us with a growing number of genome and metabolome databases to explore. Our lab is using these data to explore uncharted territories of human biology by creating exciting hypotheses to test. We have already found new mutations in genes encoding mechanosensitive proteins associated with surprising phenotypic traits from half a million individuals. We will investigate more novel areas of human biology where mechanotransduction plays unrecognized roles.



