Kwarteng, E., Li, Y., Nguyen, D., and M. Agathocleous. 2026. Stem cell function in vivo is supported by an alternative glycolysis endpoint. (bioRxiv)
*Chen, G., *Jun, J.H., Wijshake, T., Kwarteng, E.O., Li, Y., Yuan, M., Rose, J. 3rd, Li, S., Cobb, S., Serpa, W., Folger, B., Li, Y., Li, L., Chen, W., Collins, J.J. 3rd, #Wang, J., and M. Agathocleous#. (2025). Loss of vitamin C biosynthesis protects from the pathology of a parasitic infection. Proceedings of the National Academy of Sciences USA 122 :e2517730122. (PubMed)
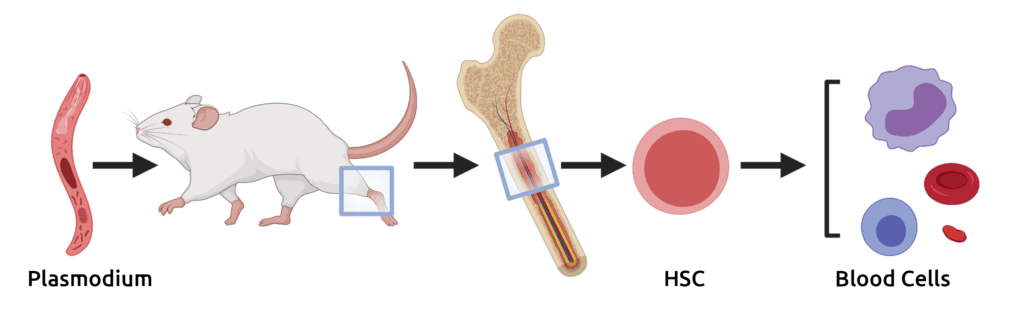
Wijshake, T., Rose, J. 3rd, Wang, J., Zielke, J., Marlar-Pavey, M., Chen, W., Collins, J. 3rd, and M. Agathocleous. (2024). Schistosome Infection Impacts Hematopoiesis. The Journal of Immunology 212, 607-616. (PubMed)
Li, Y., Hook, J.S., Ding, Q., Xiao, X., Chung, S.S., Mettlen, M., Xu, L., Moreland, J.G., and M. Agathocleous. (2023). Neutrophil metabolomics in severe COVID-19 reveal GAPDH as a suppressor of neutrophil extracellular trap formation. Nature Communications 14, 2610. (PubMed)
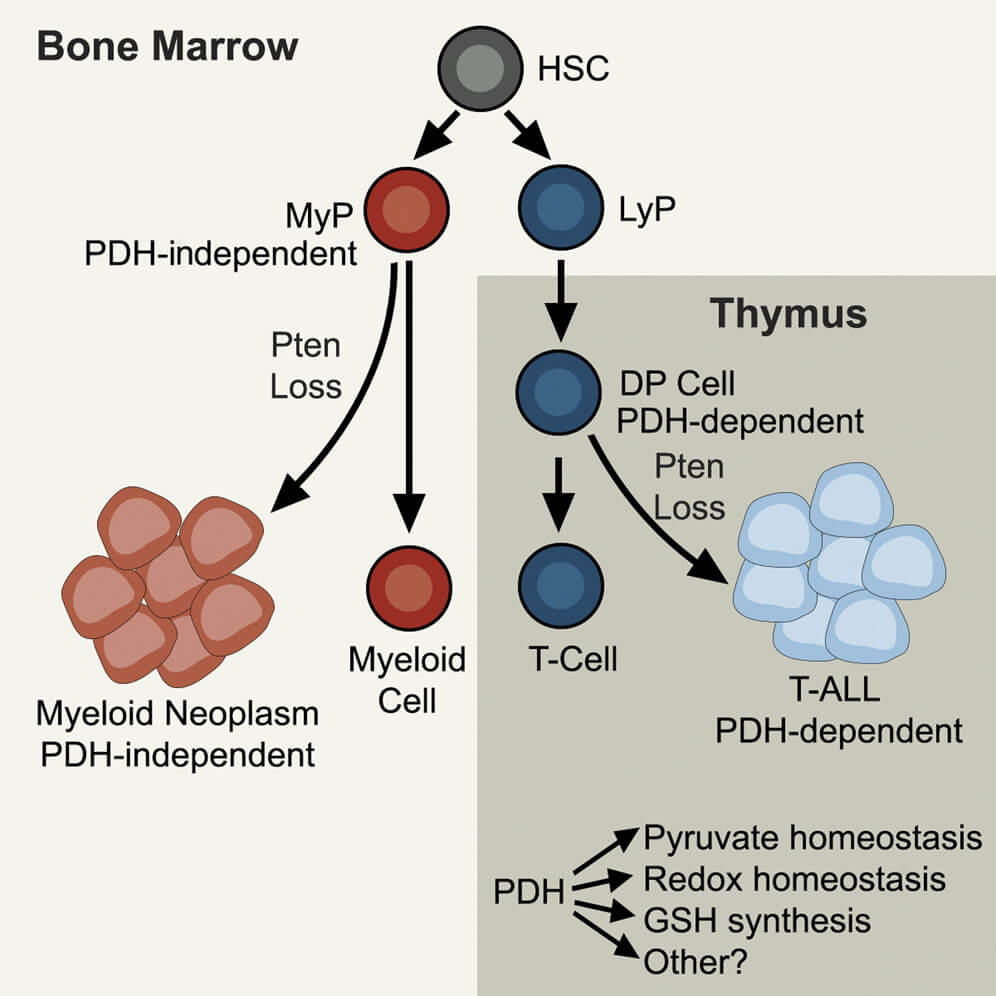
Jun, S., Mahesula, S., Mathews, T.P., Martin-Sandoval, M.S., Zhao, Z., Piskounova, E., and M. Agathocleous. (2021). The requirement for pyruvate dehydrogenase in leukemogenesis depends on cell lineage. Cell Metabolism 33, 1777-1792. (PubMed)
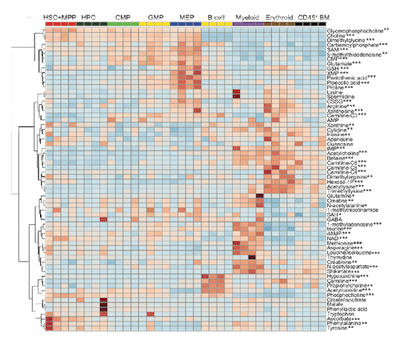
Agathocleous, M., Meecham, C.E., Burgess, R.J., Piskounova, E., Zhao, Z., Crane, G.M., Cowin, B.L., Bruner, E., Murphy, M.M., Chen, W., Spangrude, G.J., Hu, Z., DeBerardinis, R.J., and S.J. Morrison. (2017). Ascorbate regulates haematopoietic stem cell function and leukaemogenesis. Nature 549, 476-481. (PubMed)
Piskounova, E., Agathocleous, M., Murphy, M.M., Hu, Z., Mann, S., Zhao, Z., Leitch, A.M., Johnson, T.M., DeBerardinis, R.J., and S.J. Morrison. (2015). Oxidative stress inhibits distant metastasis by human melanoma cells. Nature 527, 186-191. (PubMed)
Love, N.K., Keshavan, N., Lewis, R., Harris, W.A., and M. Agathocleous. (2014). A nutrient-sensitive restriction point is active during retinal progenitor cell differentiation. Development 141, 697-706. (PubMed)
Agathocleous, M., Love, N.K., Randlett, O., Harris, J.J., Liu, J., Murray, A.J., and W.A. Harris. (2012). Metabolic differentiation in the embryonic retina. Nature Cell Biology 14, 859-864. (PubMed)
*Contributed equally
#Co-corresponding author