
Scientists at the Children’s Medical Center Research Institute at UT Southwestern (CRI) have pioneered a new method for conducting in-depth research on malignant tumors in patients, in the process discovering new complexities underlying cancer biology and overturning a nearly century-old perception about cancer metabolism.
Focusing on specific regions of tumors in patients with lung cancer — the leading cause of cancer death worldwide — Dr. Ralph DeBerardinis and his team at CRI analyzed the process by which different areas of tumors regulate the sources of energy needed for growth, and the manner in which the surrounding cellular environment influences that metabolic activity. The findings, published in Cell, may pave the way for exploiting cancer metabolism to predict disease progression and treat cancer.
“One of the compelling ideas cancer biologists have pursued for many years is that it might be possible to treat all cancers in a similar way by cutting off the fuel supply to the tumor cells — essentially starving them to death,” said Dr. DeBerardinis, Associate Professor of CRI and Pediatrics, Chief of the Division of Pediatric Genetics and Metabolism at UT Southwestern, and Director of CRI’s Genetic and Metabolic Disease Program. “But we were surprised in our study to find how much metabolic variety there was among the tumors — in some cases detecting different metabolic activities even within different areas of the same tumor — which makes it unlikely that a “starvation bullet” to treat cancers can be developed.” In addition, it appears that the metabolic preference of tumors that is determined by their genetics can be overridden by the cellular environment. Tumors — and even different regions of the same tumor — that receive a lot of blood flow were found to use many different nutrients for energy. Tumors and different areas of the same tumor that receive less blood flow were more likely to use glucose as the primary fuel.
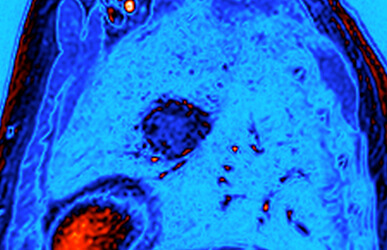
“Now that we have a much better handle on how metabolism operates in lung tumors, and where particular metabolic activities are located within a tumor, it should be easier to study exactly which activities predict and stimulate disease progression,” said Dr. DeBerardinis, who at UT Southwestern is also affiliated with the Eugene McDermott Center for Human Growth & Development, holds the Joel B. Steinberg, M.D. Chair in Pediatrics, and is a Sowell Family Scholar in Medical Research. To determine how differences in blood flow might affect tumor metabolism, the CRI research team measured the amount of blood flowing into various parts of tumors in patients by using a suite of advanced imaging techniques in collaboration with Dr. Robert Lenkinski, Professor of Radiology at UT Southwestern. The CRI team also worked with Dr. Kemp Kernstine, Professor and Chief of the Division of Thoracic Surgery at UT Southwestern, to pinpoint which regions of the tumors needed to be sampled during the removal process based on the results of the imaging investigation. Following surgical removal of the tumors, the research team performed fragment-by-fragment metabolic flux analysis to examine the metabolic changes that occurred, in collaboration with Dr. Craig Malloy, Professor and Director of the Advanced Imaging Research Center at UT Southwestern.
At UT Southwestern, Dr. Lenkinski holds the Charles A. and Elizabeth Ann Sanders Chair in Translational Research, and the Jan & Bob Pickens Distinguished Professorship in Medical Science, in Memory of Jerry Knight Rymer and Annette Brannon Rymer, and Mr. and Mrs. W.L. Pickens; Dr. Kernstine holds the Robert Tucker Hayes Foundation Distinguished Chair in Cardiothoracic Surgery; and Dr. Malloy holds the Richard A. Lange, M.D. Chair in Cardiology.
“This information will provide new opportunities to identify prognostic information and, potentially, new treatment modalities for lung cancer,” said Dr. Kernstine.
The study’s first author is Dr. Christopher Hensley, a former M.D./Ph.D. student at CRI and now an M.D. trainee at UT Southwestern. The project was supported by the National Institutes of Health, the Welch Foundation, the Cancer Prevention and Research Institute of Texas, the Canadian Institutes of Health Research, the V Foundation, and donors to the Children’s Medical Center Foundation.
About CRI
Children’s Medical Center Research Institute at UT Southwestern (CRI) is a joint venture established in 2011 to build upon the internationally recognized scientific excellence of UT Southwestern Medical Center and the comprehensive clinical expertise of Children’s Medical Center, the flagship hospital of Children’s HealthSM. CRI’s mission is to perform transformative biomedical research to better understand the biological basis of disease, seeking breakthroughs that can change scientific fields and yield new strategies for treating disease. Located in Dallas, Texas, CRI is creating interdisciplinary groups of exceptional scientists and physicians to pursue research at the interface of regenerative medicine, cancer biology and metabolism, fields that hold uncommon potential for advancing science and medicine. More information about CRI is available at: cri.utsw.edu..



